
Welcome to Evolve Med Spa!
We invite you to an aesthetic and wellness experience like no other at Evolve Medical Associates in Charlotte, NC. Our top priority is your overall wellness and natural results.
Our med spa, located in the Southpark area of Charlotte, NC, specializes in addressing your aesthetic and wellness needs through a variety of cutting edge treatments delivered with expert care. We focus on combination therapies that yield noticeable yet natural results.
From advanced laser technologies that can exponentially improve the look, tone and youthfulness of your skin to injectables and threads that soften wrinkles, add volume, and lift skin, we are excited to partner with you on the journey to a refreshed and rejuvenated appearance. Your custom treatment plan is designed with your aesthetic goals in mind and armed with our years of experience and state of the art treatments including BOTOX ® Cosmetic, Juvederm, Revanesse Versa, Halo Laser, Forever Young BBL, SkinPen Microneedling, and more.
In addition to looking refreshed on the outside, our team knows that it’s important to feel good on the inside. Ask us about our Wellness services, including Blood Panel Testing.
Contact us today and let’s begin the journey to EVOLVE.
Meet DONALD SUDY, MD
Dr. Sudy received both his undergraduate and medical degrees from the University of Washington in Seattle, WA. Following medical school, he served as a Flight Surgeon in the Air Force. during that time, he was involved with establishing the Health and Wellness Center on base as well as serving as Chief of Flight Medicine. Following his active duty commitment, he completed an Emergency Medicine residency at the University of Louisville, serving as Chief Resident in his senior year.

Quality Cosmetic Dermatology Services in Charlotte
At Evolve Medical Associates, you can enhance your appearance in an environment that’s designed to make you feel comfortable and excited to treat yourself. Our medspa offers a range of aesthetic services performed by experts. Using state-of-the-art tools, we can address all areas of your skin so you can love the way you look and feel.
- BOTOX® Cosmetic Injections in Charlotte
- BOTOX® Alternatives
- Dermal Fillers
- Vampire Facial
- VI Peel®
- SkinPen® Microneedling
- Revanesse® Versa™
- Restylane® Kysse
- SYLFIRM X RF Microneedling
BOTOX® Cosmetic Injections in Charlotte
To treat “action lines,” or wrinkles caused by repetitive motion, such as crow’s feet and forehead creases, we offer injections of BOTOX® Cosmetic. This neuromodulator works by freezing muscles to prevent the movements that cause unsightly wrinkles and an aging look. Our doctors will work with you during a personal consultation to discuss your goals and whether BOTOX® Cosmetic, or an alternative, is best for you.
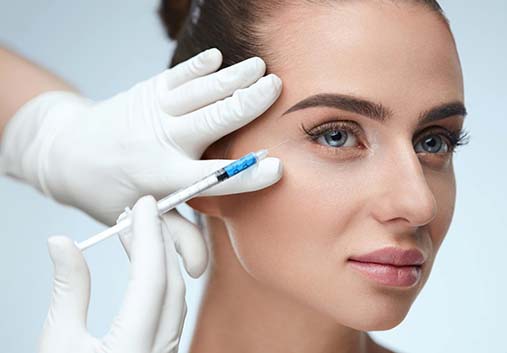
BOTOX® Alternatives
For those whose needs align with a different neuromodulator than BOTOX® Cosmetic, we offer other leading alternatives, such as Dysport®, XEOMIN®, and Jeuveau®. If you plan on using injectables long-term, XEOMIN® is a great choice because it doesn’t have any ingredients that can lead to antibody resistance. Dysport® is a thinner consistency than BOTOX® Cosmetic, meaning it spreads with ease, making it great for smooth areas like the forehead region. If you want a product that’s focused on treating frown lines, then Jeuveau® may be perfect for you.
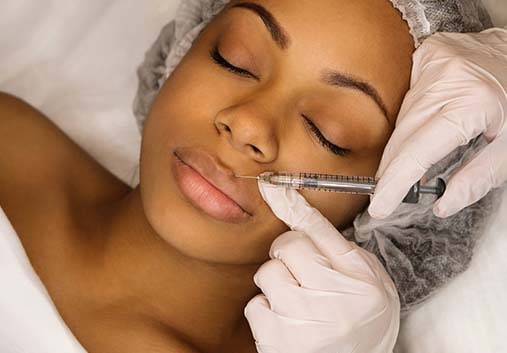
Dermal Filler
We offer a spectrum of fillers such as Restylane®, JUVÉDERM®, and RADIESSE®, to restore volume loss, renew your appearance, and enhance your features. Dermal fillers are injected under the skin to replenish fullness in areas such as the under-eye region, nasolabial folds (smile lines), and more. These products are perfect for achieving a youthful look, and can also target specific features like the jaw, cheeks, and lips to give you a contoured profile or a more luscious pout.

Vampire Facial
The vampire facial is a science-based approach to skin rejuvenation that pairs microneedling with your own body chemistry. Using microneedling, which creates micro-injuries in the skin, we’ll kickstart your body’s production of collagen and elastin — essential proteins that keep your skin firm and resilient. Then, we’ll apply platelet-rich plasma (PRP) from your own blood to the treatment area, which has been shown to boost the process further and give you dramatically refreshed skin!
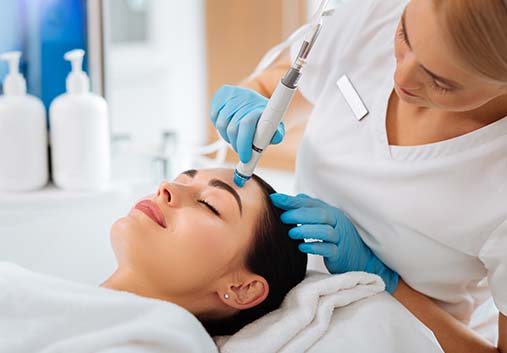
VI Peel®
To give you a radiant complexion, it’s important to slough away anything that can dull your skin’s appearance. The VI Peel® does that with safe, yet potent chemicals to strip away dirt, oil, and dead skin cells and stimulate healthy cell turnover for gorgeous results in as few as seven days. Our specialists will customize your VI Peel® to meet your needs — whether that’s reducing the appearance of sun damage, scarring, uneven skin tone, or fine lines.

SkinPen® Microneedling
Another form of microneedling that leaves you glowing and involves little downtime is the SkinPen®. This small device can precisely address specific problem areas, making it great for acne dimpling and scarring, as well as treating delicate neck skin for wrinkles. The first microneedling device to become FDA Approved, this versatile procedure is great for all skin types and tones and takes as little as three sessions to see results.
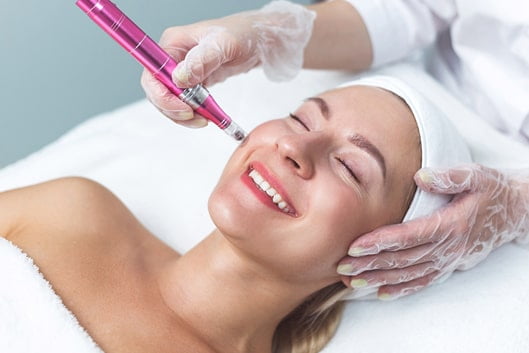
Revanesse® Versa™
Another outstanding dermal filler for facial wrinkles and fine lines is the FDA Approved Revanesse® Versa™. This hyaluronic acid filler injectable is used to help rewind aging in areas around the mouth, forehead, chin, and more. While Revanesse® Versa™ works on the skin by adding volume to areas that have started to show aging, it can also be used to create more shapely lips, cheeks, or other features for a more defined look.
Restylane® Kysse
For areas that experience a lot of movement, we offer Restylane® Kysse, a game changing dermal filler that leaves you with supple, expressive lips. This filler uses XpresHAn Technology™, which allows the particles to easily marry with your tissue for a natural, smooth movement that never looks stiff!

SYLFIRM X RF Microneedling
This FDA Approved, dual-powered procedure combines the benefits of microneedling and radiofrequency to tighten skin and treat a range of issues. By using tiny needles to puncture the skin, the body’s natural healing response is activated, stimulating collagen production for bouncy skin! SYLFIRM X also pulsates radiofrequency waves as the needles enter the skin, which further triggers skin remodeling at a deeper level for even more noticeable results.






